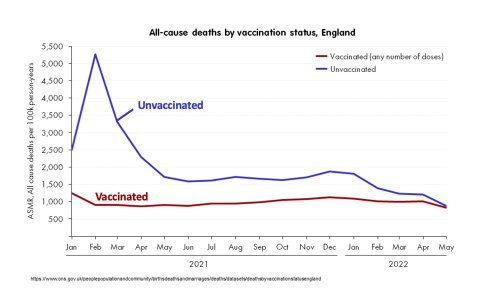
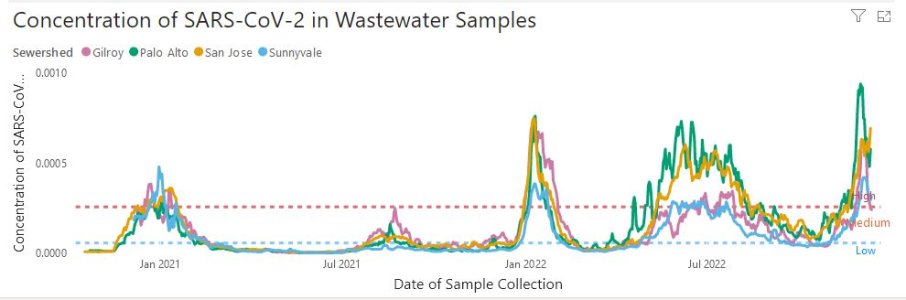
- 5,039
- 7,458
- Joined
- Jun 20, 2014
#ModernaGang winning? (Self) Reported 93% efficacy after 6 months in most recent press release
 investors.modernatx.com
investors.modernatx.com
Moderna COVID-19 Vaccine mRNA-1273: Final blinded analysis of Phase 3 COVE study shows 93% efficacy; Efficacy remains durable through six months after second dose
Moderna booster candidates demonstrate robust antibody responses to COVID-19 variants of concern in Phase 2
Dosing started in Phase 1 studies for quadrivalent seasonal flu vaccine candidate (mRNA-1010) and IL-2 mRNA program for autoimmune disorders (mRNA-6231)
Moderna has mRNA candidates in clinical development across five therapeutic areas: infectious disease, cardiovascular, oncology, rare disease and autoimmune disorders
Moderna Reports Second Quarter Fiscal Year 2021 Financial Results and Provides Business Updates | Moderna, Inc.
Moderna COVID-19 Vaccine mRNA-1273: Final blinded analysis of Phase 3 COVE study shows 93% efficacy; Efficacy remains durable through six months after second dose Moderna booster candidates demonstrate robust antibody responses to COVID-19 variants of concern in Phase 2 Dosing started in Phase 1
Moderna COVID-19 Vaccine mRNA-1273: Final blinded analysis of Phase 3 COVE study shows 93% efficacy; Efficacy remains durable through six months after second dose
Moderna booster candidates demonstrate robust antibody responses to COVID-19 variants of concern in Phase 2
Dosing started in Phase 1 studies for quadrivalent seasonal flu vaccine candidate (mRNA-1010) and IL-2 mRNA program for autoimmune disorders (mRNA-6231)
Moderna has mRNA candidates in clinical development across five therapeutic areas: infectious disease, cardiovascular, oncology, rare disease and autoimmune disorders
 …All this rush to be “in person” and “back to” for what?
…All this rush to be “in person” and “back to” for what?